MOSCOW -- In early September, as the number of new coronavirus infections in Russia was rising again, Boris Mikhailov, a graphic designer from Moscow, saw an online ad for volunteers to test a vaccine against COVID-19.
Russia’s vaccine, dubbed Sputnik-V, had been approved by state regulators the previous month, and Mikhailov decided he had to be among the participants of its Phase III trial -- the internationally recognized final stage of testing before a vaccine can be pronounced safe and effective.
“The faster they test it, the faster they’ll launch it. The faster they launch it, the faster people will stop dying,” the 53-year-old said, summarizing his reasoning in a phone interview this week. “We have to help Russian scientists by trying it ourselves.”
On the website of the Moscow city government, Mikhailov filed an application and waited for an answer. After a quick medical checkup, he was invited to a Moscow clinic to receive the first of two injections that comprise the vaccine.
Mikhailov, who said he experienced no side effects, is among 40,000 volunteers in Moscow who have helped test Sputnik-V, which was developed by Moscow’s Gamaleya Institute with funding from the state-run Russian Direct Investment Fund (RDIF). Its marketing name is a tribute to a Soviet-era triumph -- the successful launch in 1957 of the satellite Sputnik into space -- and the RDIF has not been shy about hyping the symbolism.
But two months after President Vladimir Putin announced that Russia was the first to approve a vaccine against the coronavirus, and despite much chest-thumping, the country’s daily infection rate has spiked to record levels -- exceeding 14,000 for the first time on October 14 -- and no mass production of the vaccine has begun.
“Maybe the scientists themselves have pushed back on the political push to get this thing out the door quickly,” Judy Twigg, a professor at Virginia Commonwealth University who studies post-Soviet health care, said in written comments to RFE/RL. “Or possibly it’s just taking them this long to ramp up production.”
Moscow has hardly been silent on the issue.
It has signed several licensing deals for Sputnik-V, with India set to produce over 300 million doses and Brazil to buy 50 million, and has claimed to have preliminary applications -- subject to the completion of trials -- from 40 countries totaling more than 1 billion doses. The RDIF says it will shoulder some of the legal responsibility in the event of harmful side effects.
Batches of the vaccine have also been sent to various Russian regions, earmarked for frontline workers who are prioritized under the emergency approval issued in August. Even as Sputnik-V’s Phase III trial continues, the Health Ministry says it expects to approve a second Russian vaccine by mid-October -- hoping to roll out at least one of the two approved shots nationally by January 2021.
But as health officials tout promising early results, they also face widespread skepticism among the public.
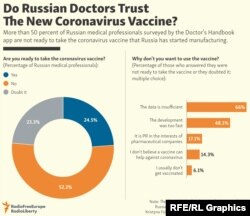
A recent survey showed that 70 percent of people in Russia would not take a coronavirus vaccine, state-run news agency RIA Novosti reported, and less than one-quarter of medical workers expressed an interest in an August poll. An opposition-backed doctors trade union has campaigned against the shot and encouraged its members to opt out.
“Our leaders should not chase their goals at the expense of your health and your freedoms, through violations of the law and by forcing your participation like guinea pigs in government experiments," the trade union, Doctors’ Alliance, said in a video showing screenshots of documents it claims were used to pressure medics to vaccinate:
The RDIF says trials of Sputnik-V are based on voluntary participation and that no one in Russia will be forced to take part. The preparation is based on two separate doses of human adenoviruses -- harmless cold viruses -- engineered to carry coronavirus proteins in quantities small enough to stimulate the production of antibodies but prevent infection.
“Based on the vaccine technology it’s derived from, it’s probably safe, and probably reasonably effective,” Twigg, the expert on Russia health care, said of Sputnik-V. “Though some of the newer technologies being used by other candidates in the U.S. and China may prove more effective.”
Besides Sputnik-V, nearly a dozen vaccines worldwide are currently undergoing late-stage trials to determine their safety and effectiveness, according to the World Health Organization.
Vaccines are usually approved after being administered to tens of thousands of volunteers; Russia’s had been given to 76 people, according to results of early testing published by Russian scientists in the medical journal The Lancet. Critics have argued that the development of Russia’s vaccine -- which received approval before undergoing Phase III trials - was expedited for political reasons to assure the country’s victory in a global race.
The RDIF’s director, Kirill Dmitriyev, has repeatedly dismissed such criticism, calling it “very unethical and very unproductive” in an interview with CNBC, claiming that Russia’s adenovirus-based vaccine is superior to those under development in the West.
The Gamaleya Institute did not respond to requests for comment. But researchers there have cited their earlier development of vaccines for Ebola and Middle East Respiratory Syndrome as a foundation that helped them forge ahead in the global race, and provided the basis for Sputnik-V. However, there is little evidence those earlier vaccines were actually used internationally in any serious way.
People are dying...We have to stop arguing and competing and unite, in order to develop a working vaccine and use it.”-- Vaccine volunteer Boris Mikhailov
An RDIF employee who was not authorized to speak on the record said Russia expects its vaccine to offer immunity lasting two years, based on results from Gamaleya’s Ebola vaccine.
There is more than geopolitics at stake. Russia had recorded 23,491 deaths as of October 15 -- though experts suspect that number may be far short of the real toll -- and its overall infection count was fourth in the world, behind only Brazil, India, and the United States. Critics warn that if the vaccine fails to prove effective it could worsen the effects of the virus -- in part by leading its recipients to abandon mask-wearing and other precautions that limit the pathogen’s spread.
For Mikhailov, such fears are misplaced. He said he trusts the Russian government’s figures and believes criticism of its vaccine are unfounded.
“People are dying across the world,” he said. “We have to stop arguing and competing and unite, in order to develop a working vaccine and use it.”
Of the 40,000 volunteers in the Moscow trial, 10,000 received a placebo -- so Mikhailov continues to wear a mask in public places and has no assurance he’s immune. In the coming weeks, he plans to test for antibodies to find out.