Vaccines are a tricky business.
Trickier, say those trying to develop and manufacture them to meet the current global COVID-19 pandemic, than tests or even medicines.
All three -- along with physical distancing and other social measures -- are vital to tracking, treating, and stamping out the current outbreak.
But the greatest challenges to widespread testing are materials and availability. And drug treatments are sensitive but also fueled by trial and error within narrow, infected populations.
Live Map: The Spread Of The Coronavirus
Vaccine work takes those potential stumbling blocks and heaps them with complex questions about efficacy, long- and short-term immunity, overreaction, and potentially deadly reactions in otherwise healthy people, to name a few.
Not to mention the feat of eventually providing billions of people with the vaccinations against a pathogen like the new coronavirus, which has infected 2.5 million people worldwide, killing more than 180,000, since emerging in central China five months ago.
"Vaccine development is always more cumbersome and longer, because you have to vaccinate large numbers of people -- therefore safety is very important and volume is very important," Paul Stoffels, chief scientific officer at pharmaceutical giant Johnson & Johnson, told a COVID-19 event organized by the European Federation of Pharmaceutical Industries and Associations (EFPIA) on April 20.
Most experts suggest the world shouldn’t expect widespread administration of a COVID-19 vaccine before early 2021 despite unprecedented levels of cooperation and sharing between academics and the pharmaceutical industry, regulatory shortcuts, and a leap into testing on humans that includes previously untested technologies.
Multiple Paths
Dozens of companies, laboratories, or individual researchers claim to be working on one or more potential COVID-19 vaccines.
But even the most advanced are still in their early stages.
Vaccine development has historically taken around 10-15 years. Virologists note that there is still no licensed vaccine against HIV, the retrovirus identified in the early 1980s as the cause of AIDS.
But outbreaks of coronaviruses like Ebola, SARS, MERS, and Zika in the intervening decades have helped speed the development of antivirus vaccine development.
A group of top virologists wrote in the New England Journal Of Medicine on March 30 that the need to "rapidly develop a vaccine against SARS-CoV-2 comes at a time of explosion in basic scientific understanding."
They said that includes areas such as "genomics and structural biology."
RFE/RL's Coverage Of COVID-19
Features and analysis, videos, and infographics explore how the COVID-19 pandemic is affecting the countries in our region.
In that environment, it is essential that candidate vaccines try to harness everything from vector- and protein-based vaccines to innovative ways to spark an immune system into action with simulated snippets of genetic material from a target virus.
As one pharmaceutical executive put it, "we don't know yet which one will get the best immunity."
Most vaccines use a dead or otherwise deactivated form of the virus -- like the smallpox vaccine that was introduced more than 200 years ago -- or insert the target virus's genetic material into a harmless virus to cue the body to produce antibodies.
Another approach could be to repurpose existing vaccines to reduce, even briefly, the chances of infection. But there is little evidence so far that deploying something like the oral poliovirus vaccine (OPV) could ward off SARS-CoV-2.
Competitive Science
Still, many researchers hope for rapid progress on COVID-19 in a new form of vaccine, known as mRNA, in which a bit of messenger genome that codes for a virus is used to spark an immune response.
No mRNA vaccine has ever been approved for widespread use on humans. But they're now being tested in the current crisis, on animals and people.
"Basic science is best when it's competitive," Seth Berkley, chief executive of the global vaccine alliance GAVI, told The Economist's Babbage podcast recently.
Berkley said development of 140 to 150 vaccines is now "moving forward."
"That's great, but we can't take 150 vaccines into large-scale clinical testing and manufacturing. So...those need to be downregulated based upon appropriate science, based upon the appropriate animal models, the target product profile."
Testing On People
So-called preclinical testing on animals began within weeks of the publication of the SARS-CoV-2 genome in January, and usually include "challenging" animals like ferrets, monkeys, and mice with the related, pneumonia-like disease.
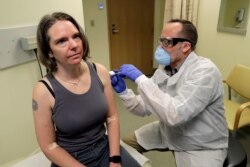
Sarah Gilbert, a professor of vaccinology at Oxford University, surprised many last week when she said her team hoped to start clinical trials on humans of a candidate vaccine -- testing a very small number of healthy people for dangerous effects -- by the end of this month.
But while testing on human subjects is a landmark, it is important to distinguish between small-sample testing -- such tests on patients are already under way in the United States, Germany, and China, for instance -- and the battery of further tests for would-be vaccines to ensure safety and assess effectiveness.
About one in three experimental drugs never make it through this so-called Phase I testing, clinical trials monitor CenterWatch says.
Another third of experimental drugs are usually winnowed out in Phase II studies. Those can last from months to years and include hundreds of patients.
Once a vaccine advances to Phase III studies, it might be administered to thousands of patients. But such research frequently takes several years.
The top U.S. infectious diseases expert, Anthony Fauci, said in March that the first phase of trials on humans started there at "record speed" and that a COVID-19 vaccine is "an urgent public health priority."
But Fauci remains guarded about the timeline for a best-case scenario, saying it won't be "in the immediate future."
"You've got to make sure you have something that's safe and that works," Fauci told the Journal Of The American Medical Association about two months after the vaccine race began in earnest with the publication of the SARS-CoV-2 genome.
"It's going to take a year to a year-and-a-half to find out if [any potential vaccine] even works," he said.
The World Health Organization (WHO) has organized a group of "scientists, physicians, funders, and manufacturers" from around the world who are urgently seeking a COVID-19 vaccine.
They convene regularly to encourage a continuation of "efforts to strengthen the unprecedented worldwide collaboration, cooperation and sharing of data already underway."
A Time For Sharing
Many pharmaceutical companies and researchers hope regulatory bodies will continue to streamline applications and approvals.
They also want those working on new vaccines to share more information and even incorporate similar data to speed testing and other phases of development.
"It's amazing how the regulators work with us," said Stoffels, whose company has spent decades on possible vaccines for HIV, Ebola, and more recently Zika.
Stoffels said "deep interaction" with groups like the U.S. Food and Drug Administration (FDA) and its European counterparts "cut three months out of the development program from the beginning" thanks to repurposing safety data from other platforms and applications.
"There is no time for not sharing," Stoffels said. "If we learn something, nobody should repeat that experiment if it didn't work out."