The key to curtailing the coronavirus pandemic ravaging the globe might well be a 15-centimeter-long stick, made of plastic or wood, with a small cotton bud on the end.
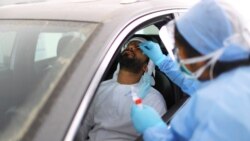
Inserted in your nose until it brushes the back of your upper throat, the swab is used to gather cells whose DNA can then be analyzed and tested for the presence of SARS-CoV-2, the scientific name for what is widely called the coronavirus.
It's not the most pleasant sensation. It is, however, one of most effective and efficient ways to test large numbers of people, experts say, and to figure out who should be quarantined, where the virus is hitting hardest, and who might be immune.
While some countries, like South Korea, have drawn plaudits for rolling out wide-scale testing of their populations, and have gotten ahead of the curve for containing coronavirus, others have stumbled. That includes Italy and Spain, two of the hardest-hit countries, as well as the United States.
Here’s what you need to know about testing.
How are the tests done?
There are two tests currently in use. The lesser-known one involves drawing blood and then testing for antibodies to the coronavirus. Antibodies are the proteins in a person's body that are activated in response to a person acquiring a disease, be it bacterial -- pneumonia, for example -- or viral, such as influenza.
Antibody-based tests, also known as serological tests, determine whether a person has had a certain disease. That typically only occurs several days after a person has been infected, whether or not they showed symptoms. That makes these tests less useful for identifying cases when time is of the essence -- when a disease is spreading rapidly through a population.
For that reason, the most important test at present for the coronavirus is known as a "reverse transcriptase polymerase chain reaction" test -- that's the one with the swab in your nose.
The test kits distributed by the World Health Organization (WHO) or the U.S. Centers for Disease Control and Prevention both use this method, as do many of the tests that are rapidly being produced by private laboratories.
The test is conducted by having a cotton-tipped swab inserted in your nose, until it reaches the back of your upper throat. For most people, it's a decidedly unpleasant sensation that often causes a person to gag.
With microscopic cells attached to the cotton tip, the swab is then inserted into a vial for safekeeping and transported to a laboratory. At the lab, workers conduct a molecular test that looks for fragments of the genome of the coronavirus. If the virus is present in great quantities, it can quickly be detected.
Why is testing for coronavirus quickly so important?
One of the reasons the coronavirus can spread fast and far is that you can have the virus for several days without exhibiting any symptoms -- and during that time, you can unknowingly transmit the virus to multiple other people.
The problem is that the coronavirus is so new, and spread so quickly, that there weren't enough tests, and not enough places to verify the tests. The explosion of cases -- in Italy, in Spain, in Britain, in the United States -- quickly dwarfed the ability of those countries' health-care systems to test for the virus.
Live Map: The Spread Of The Coronavirus
And that doesn't take into account countries whose public health systems were creaky to begin with: Iran, for example.
"One of the issues that we have with this particular virus...is that we still don't have an observational way of knowing who is infected and who isn't," said Andrew Preston, a professor of biology and biochemistry at the University of Bath, in England. "So we can't rely on symptoms alone, because it takes a little while for those symptoms to start to show, even after someone's picked up the virus.
"So if we want to do any kind of determination of who has the virus, who has the infection, then we need to test so that's why testing is so important," he said in an interview with RFE/RL.
Epidemiologists and medical experts say the lack of rapid, widespread testing has hindered some countries' responses.
As of March 29, the United States, whose population is 330 million, had performed tests on 1 in 366 people, roughly. By comparison, Italy, whose population is about 60 million and is one of the worst-hit countries, has tested 1 in 133 people, and much of that came after a surge in testing as Italians already with the disease overwhelmed the country's hospitals.
Russia, meanwhile, had conducted 343,500 tests as of March 30. With a population of 144 million, that means only 1 in 419 people had been tested.
Russia's efforts to ramp up testing were stymied by existing protocols: relying on a single Siberian laboratory to do second, confirmation tests of preliminary results. And some advocates have said the test that had been in wide use was flawed, producing false negatives. The government has since shifted tactics, and is doing wider and more rapid testing.
Most affected countries are ramping up testing to get a wider segment of their populations, so those testing rates will improve.
Why are some countries able to do it quickly, while others are struggling?
Though considered highly accurate, the nasal-swab test does take some time to conduct: from the moment a person's nose is swabbed, to the time that a lab can do the test, and then notify the person of the test results. And that's under the best of circumstances.
Some countries have pushed to quicken testing protocols: drive-through testing is now appearing in some places, where people can be tested while seated in their cars.
South Korea has successfully utilized a telephone-booth type of system: People step into glass booths and are tested quickly and safely. This also minimizes the possibility of a medical worker being infected while conducting a test.
South Korea, which has reportedly tested around 1 in 250 people, has also benefited from its previous experience with SARS, the respiratory illness that also emerged in China in the early 2000s, according to Preston.
That disease infected fewer than 8,500 people worldwide, and killed an estimated 774 people, according to the WHO.
"My feeling is that [South Korea] had better capacity for testing early on," he said. "And also they really, really triggered that tracing, you know, identification and contact tracing of those first cases very quickly."
With a large, well-funded, modern health-care system, and some of the world's best medical universities, the United States should have been in a good position to test quickly and widely.
That didn’t happen for a variety of reasons, experts say, including regulatory delays and mixed messages about the scope of the problem from the White House and elsewhere.
"Testing is the biggest problem that we're facing," said Peter Slavin, president of Massachusetts General Hospital in Boston.
"The idea that you can suddenly go from having a capacity where you can run hundreds of tests today, which is kind of all you would need to do for most diseases during normal situations, to doing tens of thousands or even hundreds of thousands of tests a week. I mean, that's difficult," Preston told RFE/RL. That's "because these tests have to be performed properly, particularly if you're going be making crucial decisions on who's OK to continue working in the hospital based on these."
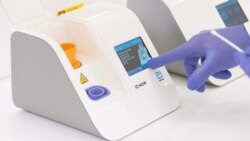
Several private companies around the world, meanwhile, are rushing to speed up the time it takes to get the actual lab result.
The U.S. company Abbott Laboratories last week won regulatory approval for a coronavirus testing system that can tell if someone is infected in as little as five minutes. The system is also small and portable.
Swiss pharmaceutical giant Roche also won U.S. approval last week for a test that provides results in three hours.
Faulty testing has also been a problem. Spain, Georgia, and Turkey are among the countries that obtained a coronavirus test from a Chinese company, but later discovered the tests were defective, having a high rate of error.
"There's huge questions arising as to what...level are we prepared to go to protect ourselves," Preston said. "So I think we need to rethink our global infrastructure for dealing with pandemics.
"We've been predicting this for so many years, but it's remained sort of a theoretical exercise to a large degree, and clearly not anymore," he said.